Endovascular Abdominal Aortic Aneurysm Repair (EVAR)
78-year-old male with enlarging abdominal aortic aneurysm (AAA).

Figure 1.
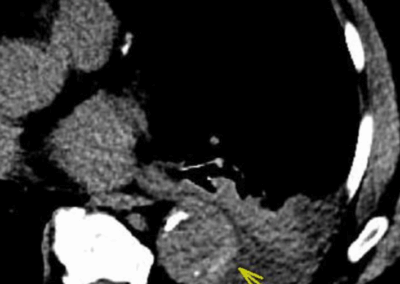
A. Surveillance axial CTA image showing 5.3 cm AAA (arrowheads) with moderate mural thrombus. The aneurysm had increased more than 5 mm over previous 12 months.
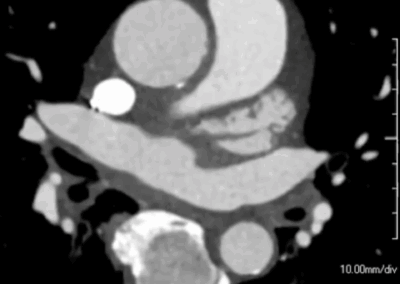
B. Post-procedural CTA after aorto-biiliac endograft placement (arrow) shows exclusion of the aneurysm with no endoleak.
CTA injection parameters:
Scan time: 4 sec
Diagnostic (“trigger”) delay: 7 sec
Bolus triggering : supra celiac aorta
Tube energy: 120 kVp
Contrast concentration: 370 mgI/mL
Contrast volume: 90 mL
Contrast flow rate: 4.8 mL/sec
Saline flush volume: 30 mL
Saline flow rate: 4.8 mL/sec

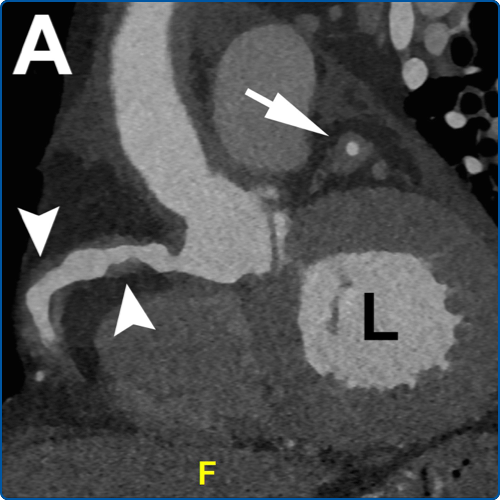
Figure 2. A. 3D Volume-rendered images before and after EVAR. A. Pre-procedural CTA shows fusiform AAA . Thrombus seen in Figure 1 is not rendered by these opacity transfer settings. Patent inferior mesenteric artery (IMA) is noted arising from the aneurysm (arrow). B. Post-EVAR CTA shows successful placement of device with proximal fixation just below the renal arteries (arrowheads) and distal fixation in the common iliac arteries (*). The IMA is no longer patent (arrow), branches fill via SMA territory via the arc of Riolan.
All images, protocols, and outcomes are courtesy of Dr. Richard Hallett, Northwest Radiology, Indianapolis, IN.
Individual results may vary.
Most Recent Cases
![]()
IMPORTANT SAFETY INFORMATION | INDICATIONS AND USAGE
EmpowerCTA®+ Injector System
Caution: Federal law restricts this device to sale by or on the order of a physician.
INDICATIONS:
The EmpowerCTA®+ Injector System is indicated for the vascular administration of contrast and flushing media in conjunction with computed tomography (CT) scanning of the body with an optional interface to a CT scanner and an optional calculator for glomerular filtration rate (GFR).
INDICATIONS AND USAGE | IMPORTANT SAFETY INFORMATION
EmpowerCTA®+ Injector System
Caution: Federal law restricts this device to sale by or on the order of a physician.
INDICATIONS:
The EmpowerCTA®+ Injector System is indicated for the vascular administration of contrast and flushing media in conjunction with computed tomography (CT) scanning of the body with an optional interface to a CT scanner and an optional calculator for glomerular filtration rate (GFR).
Protocols have been independently developed and are not intended as medical advice. Bracco and Bracco Injeneering S.A. shall not be responsible for any physicians’ reliance on these or any other products.
CONTRAINDICATIONS:
The EmpowerCTA®+ Injector System is not intended for use as a long-term infusion pump, nor is it intended to be used to inject any agents other than contrast or flushing media. Do not attempt to use the injector for any other purpose (such as chemotherapy or drug infusion). The EmpowerCTA+ Injector System should not be used to inject substances into nonvascular body cavities. Any applications of the EmpowerCTA+ Injector System other than those described in its Operator Manual are inappropriate and should not be attempted.
Not all products are available in all global markets.
EmpowerCTA+ is distributed by Bracco Diagnostics Inc.; 510 Carnegie Center, Suite 300, Princeton, NJ 08540 USA; Phone: (800) 631-5245; Fax: (609) 514-2424; Customer Service: 1-877-BRACCO 9 (1-877-272-2269); Scientific Information: 1-800-257-5181 (Option 2); EmpowerCTA+ Email: empowerinfo@diag.bracco.com; Website: https://smartinject.com/ct-solutions/
EmpowerCTA+ is manufactured by Bracco Injeneering S.A., Avenue de Sevelin 46, CH – 1004 Lausanne, Switzerland. https://www.bracco.com/
EmpowerMR® Injector System
Caution: Federal law restricts this device to sale by or on the order of a physician.
INDICATIONS:
The EmpowerMR® Injector System is indicated for vascular administration of contrast media and flushing media in conjunction with magnetic resonance imaging (MRI).
Protocols have been independently developed and are not intended as medical advice, and Bracco shall not be responsible for any physicians' reliance on these or any other protocols.
Bracco Injeneering S.A. reserves the right at any time and without notice, to change the specifications and features described herein, or to change the production or adjust the product described.
Not all products are available in all global markets.
EmpowerMR is distributed by Bracco Diagnostics Inc.; 510 Carnegie Center, Suite 300, Princeton, NJ 08540 USA; Phone: (800) 631-5245; Fax: (609) 514-2424; Customer Service: 1-877-BRACCO 9 (1-877-272-2269); Scientific Information: 1-800-257-5181 (Option 2); Website: https://smartinject.com/mr-solutions/
EmpowerMR is manufactured for Bracco Injeneering S.A., Avenue de Sevelin 46 CH – 1004 Lausanne, Switzerland. https://www.bracco.com/
About Bracco Injeneering S.A.
Bracco Injeneering S.A., located in Lausanne, Switzerland, is the most recently added Business Unit of Bracco Imaging. Bracco Injeneering is committed to developing the best-in-class, integrated injection solutions with a strong heritage in research and innovation. It provides quality solutions for state-of-the-art radiology centers, offering proven injection technology, built on Bracco Imaging’s expertise.
Thanks to this strengthened product portfolio, including the EmpowerCTA®+ Injector System and EmpowerMR Injector System Bracco Imaging will be able to focus on constant innovation, not only for devices but also for software development and data management.
EmpowerCTA, EmpowerMR, IRiSMR, and NEXO are registered trademarks of ACIST Medical Systems, Inc.
FastLoad is a trademark of ACIST Medical Systems, Inc.
NEXO® Contrast Management System
Intended Use:
The NEXO® Contrast Management System is a server-based application intended to be used as a data management and visualization system. The NEXO Contrast Management System can read, store, share, compare/relate, as well as graphically display data and injection programs coming from RIS, PACS, and multiple enabled EmpowerCTA® Injector Systems. This device provides users with record lists, graphics, and reports about data and performances.
This device is intended for retrospective and statistical data management and visualization. It is not intended for real-time data visualization, diagnostic applications, or for any other use for which the device is not indicated. This device is only to be operated by and under quasi-continuous supervision of qualified and trained staff in an appropriately licensed healthcare facility.
The NEXO Contrast Management System is compatible with the EmpowerCTA Injector System version 8.0, and EmpowerCTA®+ Injector System version 10.0 and higher.
Not all products are available in all global markets.
NEXO is distributed by Bracco Diagnostics Inc.; 510 Carnegie Center, Suite 300; Princeton, NJ 08540 USA; Phone: (800) 631-5245; Fax: (609) 514-2424; NEXO Customer Service: 1-877-BRACCO 9 (1-877-272-2269); NEXO questions: Nexo.Answers@diag.bracco.com; NEXO U.S. Technical Service and Support: Phone: 888-670-7701; Fax: 609-514-2448; NEXO Email: customer.support@acistmedical.com; Website: https://smartinject.com/digital-solutions
NEXO is manufactured by ACIST Medical Systems, Inc., 7905 Full Road, Eden Prairie, MN 55344 USA.
NEXO and EmpowerCTA are registered trademarks of ACIST Medical Systems, Inc.
ulricheasyINJECT Max 3™ (the Bracco-branded Max 3™, a Rapid Exchange and Syringeless MR Injector System) is distributed by Bracco Diagnostics Inc.
Indications for use
ulricheasyINJECT Max 3 (XD 10180) is a contrast media management system that is indicated for the controlled, automatic administration, on the venous side, of contrast media and saline (NaCl), to human subjects undergoing diagnostic examinations in magnetic resonance (MR) applications.
ulricheasyINJECT Max 3 (XD 10180) is specifically indicated for use in MRI procedures for the delivery of the following contrast media:
• Gadobutrol Injection in single-dose (SD) container or Imaging Bulk Package (IBP)
• Gadopiclenol Injection in SD container or IBP
• Gadobenate dimeglumine Injection in SD container
• Gadoterate meglumine Injection in SD container
Easy-Click-Cassette – flex Max 3 is used for a maximum time of twenty-four (24) hours or a maximum of 96 bottles of contrast media, whichever comes first.
Use time expiration per SD container is a maximum of four (4) hours, unless otherwise stated by the contrast media labeling.
Use time expiration per IBP or saline container is a maximum of twenty-four (24) hours, unless otherwise stated by the media labeling.
Spike for MRI disposable is for single-bottle use only and must be discarded with the media container. The Patient tubing must be discarded after each patient procedure.
ulricheasyINJECT Max 3 (XD 10180) is to be used only by and under quasi-continuous supervision of trained healthcare professionals in an appropriate licensed healthcare facility, in a room designated for radiological procedures that involve intravascular administration of contrast agent.
The ulricheasyINJECT Max 3 (XD 10180) is not intended for injection of contrast media (CM) for high-pressure angiography.
Contraindications
ulricheasyINJECT Max 3 injectors are not intended for the administration of contrast medium during high-pressure angiography or other applications that do not comply with the intended use.
The injector is not protected against the effects of defibrillation. Before a defibrillator is used, the patient must be disconnected from ulricheasyINJECT Max 3 injector.
Do not add any disposables (i.e. connector tubing or valves) to the ulricheasyINJECT Max 3 disposables or in conjunction with the patient tubing that are not provided by ulrich medical.
No valves or other connectors may be placed in-line between the patient tubing and the patient cannula. The disposables identified in this IFU are designed, manufactured, and tested for connection with cannulas for pressure injections.
Do not use ulricheasyINJECT Max 3 injectors with any other contrast media (other than those described in this IFU). Any other contrast media are inappropriate and should not be used.
Do not operate the injector and terminal, including any accessories, in potentially explosive atmospheres or in the vicinity of combustible materials (especially anesthetic drugs, detergents, and oxygen-enriched environments).
ulricheasyINJECT Max 3 is manufactured by ulrich GmbH & Co. KG.
ulrich medical is a registered trademark of ulrich GmbH & Co. KG.
ulricheasyINJECT Max 3 is a trademark of ulrich GmbH & Co. KG.
ulricheasyINJECT Max 3 is distributed as the Bracco-branded Max 3, a Rapid Exchange and Syringeless MR Injector System, by Bracco Diagnostics Inc.; 510 Carnegie Center, Suite 300, Princeton, NJ 08540 USA; Phone: (800) 631-5245; Fax: (609) 514-2424; Customer Service: 1-877-BRACCO 9 (1-877-272-2269); Scientific Information: 1-800-257-5181 (Option 2); Website: https://smartinject.com/max3/
All other trademarks and registered trademarks are the property of their respective owners.